 
Understanding Bohr's model requires some knowledge of electromagnetic radiation (or light).īohr's key idea in his model of the atom is that electrons occupy definite orbitals that require the electron to have a specific amount of energy. Its value is obtained by setting n 1 in Equation 6.38: a0 40 2 mee2 5.29 × 1011m 0.529Å. The radius of the first Bohr orbit is called the Bohr radius of hydrogen, denoted as a0. In 1913, the Danish physicist Niels Bohr proposed a model of the electron cloud of an atom in which electrons orbit the nucleus and were able to produce atomic spectra. The electron’s speed is largest in the first Bohr orbit, for n 1, which is the orbit closest to the nucleus. Underappreciated fact about Bohrs model: it was his explanation of the appearance of helium in starlight that was unprecedented it was what convinced. The energy required to remove the second electron is 54.4 eV, as would be expected by modeling it after the hydrogen energy levels. This image of galaxy cluster SMACS 0723 and its surroundings was the first image released from the James Webb. The energy required to remove one of them is the highest ionization energy of any atom in the periodic table: 24.6 electron volts. James Webbs too massive galaxies may be even more massive. These difficulties cast a shadow on the planetary model and indicated that, eventually, it would have to be replaced. Helium Energy Levels The helium ground state consists of two identical 1s electrons. Furthermore, Rutherford's model was unable to describe how electrons give off light forming each element's unique atomic spectrum. The two-electron model described here can be simplified to a one-electron problem because the. So the ionisation energies of H, H e X , L i X 2 are respectively 1, 4, 9. If the electron circling the nucleus in an atom loses energy, it would necessarily have to move closer to the nucleus as it loses energy, and would eventually crash into the nucleus. Bohrs theory tells us that the ionisation energy for one-electron atoms varies with the square of the nuclear charge, i.e. This is, after all, how we produce TV signals. It was already known that when a charged particle (such as an electron) moves in a curved path, it gives off some form of light and loses energy in doing so. Unfortunately, there was a serious flaw in the planetary model. Electron 1 starts at (r1,0,0), while electron 2 starts at (-r1,0,0).\): Niels Bohr with Albert Einstein at Paul Ehrenfest's home in Leiden (December 1925). 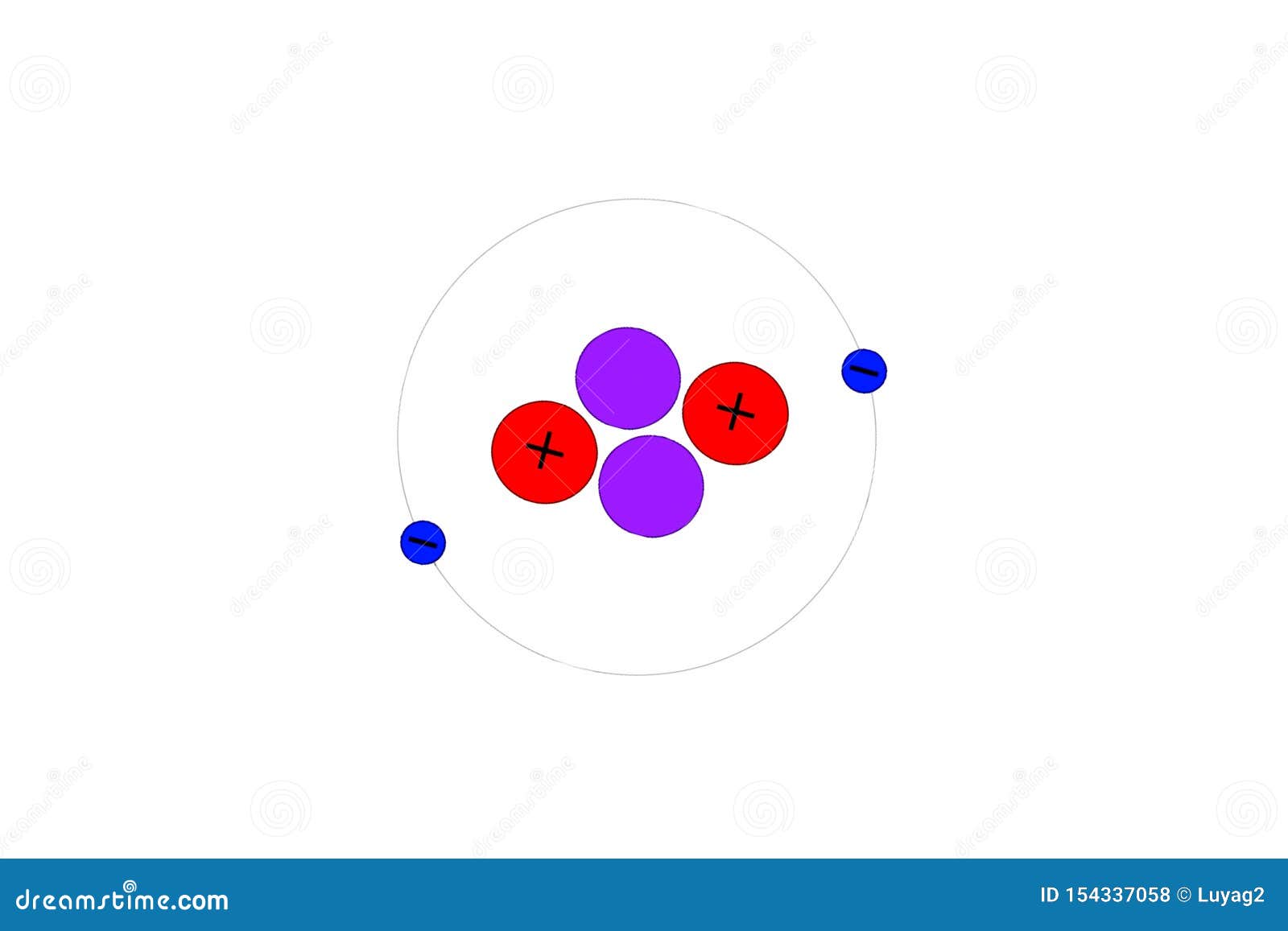
This figure shows one quater of the orbitals. 
Two same-shaped orbital planes of electrons are perpendicular to each other. Atoms are made up of sub-atomic particles. I grant anyone the right to use this work for any purpose, without any conditions, unless such conditions are required by law.Ĭlick on a date/time to view the file as it appeared at that time. Answer Atomic Models Questions after you have finished. In some countries this may not be legally possible if so: Draw Bohr atomic models for each of the atoms using your Periodic Table. S1-02-05 Assemble or draw Bohr atomic models for the first 18 elements and. I, the copyright holder of this work, release this work into the public domain. The Bohr model: Journey to find structure of atoms. 1 Balloon of Xenon 1 Balloon of Air 4 Balloons of Helium Periodic Table. Licensing Public domain Public domain false false The orbital length is proved to be just one times de Broglie's wavelength. By a computational method, we calculate the Coulomb force among the particles and the number of de Broglie's waves contained in the short segment at short time intervals. Electron 1 starts at (r1,0,0), while electron 2 starts at (-r1,0,0). This figure shows one quarter of the orbitals. But this model could not explain the experimental value of the Helium ground state energy. Two same-shaped orbital(not circular) planes of electrons are perpendicular to each other. In Bohr model Helium, the two electrons are moving on the same orbital. English: Bohr model of Helium _ (This model is just consistent with the experimental value of helium ground state energy). According to Bohrs model.the radius of the second orbit of helium atom is: 0.53A 1.06A 2.12A 0.265A rnZn2×0.529A.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
